
Improving Laboratory Management
This Website Is Intended To Provide Medical Ebooks For Free Download By Doctors & Medical Students. CLICK HERE FOR MEDICAL BOOKS FREE DOWNLOAD FOR THOSE MEMBERS WITH BLOCKED DOWNLOAD LINKS. Numerous other software packages are available, including based data analysis with Excel. GET IT FREE HERE. When you want the best professional and cost effective software for lab reporting - get PathoPlus - The ultimate software for Lab Reporting. PathoPlus is an industry-standard complete Laboratory Information Management System (LIMS) which will automate your lab and facilitate your organization with fast & smooth workflow.
One way to meet these challenges head on is to use a laboratory quality management software system (QMS). A quality management software system that is automated and connects all departments is essential for a regulated or ISO-compliant company. A QMS or a TQM (total quality management) system can connect each department in a laboratory with other departments. Automated routing, with escalation, ensures the rapid responses to inputs needed from other departments. By building quality into products as opposed to forcing QA to bear the burden of the responsibility, everyone (engineering, regulatory, QA, manufacturing, sales and marketing) wins.
When is it Time to Get a Laboratory Quality Management Software System (QMS)?
The best scenario would be that the day the lab opens, a laboratory QMS is in place. Document control is essential to the safety and efficacy of a lab. Additionally, in order to comply with accreditation, laboratories must establish an effective document control system. Each document must be created, updated, approved, and archived in specific ways, or a laboratory will have deficiencies on the accrediting agency’s checklist.
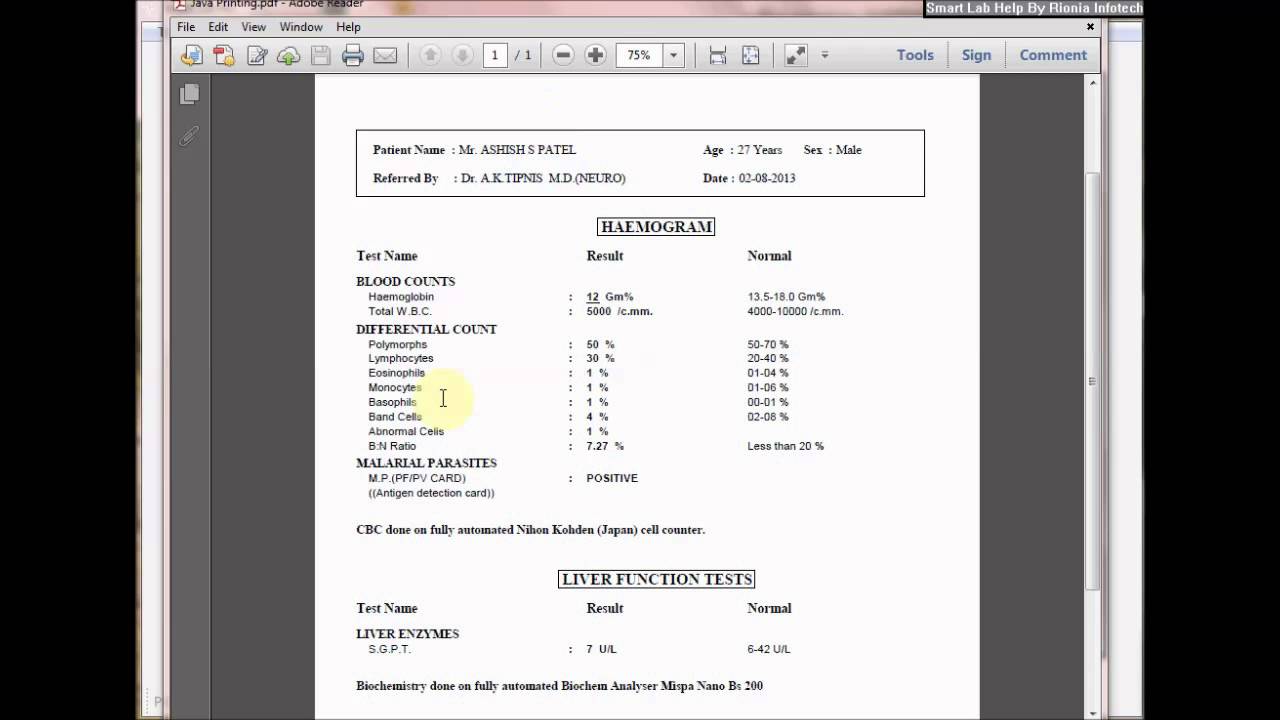
Many laboratories keep their policies and procedures in physical binders, trying to keep up with the endless paper trail of revised standard operating procedures and policies. There are usually multiple copies of these binders in different parts of the lab, making updating these documents very time-consuming and error-prone.
The best time to get a laboratory quality management software system may be before accreditation rolls around. The accreditation process includes a review of management systems and an assurance that a laboratory has a quality system that is documented and fully operational. An electronic document control system provides this kind of assurance.
Improving Laboratory ManagementOne way to meet these challenges head on is to use a laboratory quality management software system (QMS). A quality management software system that is automated and connects all departments is essential for a regulated or ISO-compliant company.
A QMS or a TQM (total quality management) system can connect each department in a laboratory with other departments. Automated routing, with escalation, ensures the rapid responses to inputs needed from other departments. By building quality into products as opposed to forcing QA to bear the burden of the responsibility, everyone (engineering, regulatory, QA, manufacturing, sales and marketing) wins. When is it Time to Get a Laboratory Quality Management Software System (QMS)?The best scenario would be that the day the lab opens, a laboratory QMS is in place. Document control is essential to the safety and efficacy of a lab. Additionally, in order to comply with accreditation, laboratories must establish an effective document control system. Each document must be created, updated, approved, and archived in specific ways, or a laboratory will have deficiencies on the accrediting agency’s checklist.Many laboratories keep their policies and procedures in physical binders, trying to keep up with the endless paper trail of revised standard operating procedures and policies.
There are usually multiple copies of these binders in different parts of the lab, making updating these documents very time-consuming and error-prone.The best time to get a laboratory quality management software system may be before accreditation rolls around. The accreditation process includes a review of management systems and an assurance that a laboratory has a quality system that is documented and fully operational. An electronic document control system provides this kind of assurance. How Can MasterControl’s Laboratory Quality Management System Benefit You?MasterControl’s Laboratory QMS is able to handle all five parts of the quality management system described above. The software helps sustain a laboratory’s compliant status, year in and year out.

